Pure NMN powder formulation trends: bioavailability and delivery tech
Mar 19, 2026
Trends in the preparation of pure NMN powder show big steps forward in improving bioavailability and improving transport technology. Modern formulation strategies focus on getting around traditional absorption barriers by using new ways to encapsulate, engineer particle size, and deliver drugs specifically. Because of these improvements, companies can now make nicotinamide mononucleotide products that work better because they can be taken in by cells better and keep working for longer. This meets the growing need for high-performance anti-ageing vitamins around the world. Nicotinamide Mononucleotide (NMN) powder has become an important ingredient in the anti-ageing and longevity fields because it helps the NAD⁺ biosynthesis. People who buy things for businesses, like buying managers, original equipment manufacturers (OEMs), distributors, and formulation engineers, know how important it is to understand new bioavailability and delivery methods in order to find high-performance NMN powders. This in-depth study shows how important it is to come up with new formulations and find the best ways to send products so that they work better and help companies stay ahead in the global supply lines. The nutrition business is still seeing NAD+ precursor demand grow at a rate that has never been seen before. Market research shows that consumers are becoming much more aware of how to improve their cellular health. Manufacturing partners have to deal with tricky recipe problems while also following strict quality standards and rules in a number of different foreign markets.
Understanding Pure NMN Powder and Its Bioavailability Challenges
Pure NMN powder, which is a key NAD+ precursor, is chemically essential for cellular energy metabolism and studies on ageing. This is why health and anti-ageing companies all over the world want to buy it. The molecular structure of nicotinamide mononucleotide makes it a great ingredient for advanced supplement recipes because it supports cell regrowth in ways that no other ingredient can.
Oral absorption, on the other hand, faces big problems that make products less successful and less competitive in the market. Manufacturers who want uniform treatment results face big problems, such as low absorption rates, stability issues in different environments, and different suppliers' variations in purity levels.
Molecular Absorption Barriers
Nicotinamide mononucleotide uptake is limited by physiological hurdles that make traditional oral delivery means less effective. There are changes in pH, risks of enzyme breakdown, and competing absorption routes in the gastrointestinal environment that make it less bioavailable overall. According to research, NMN chemicals that are not protected may break down at rates higher than 60% during normal digestion processes.
Solubility and Particle Size Optimisation
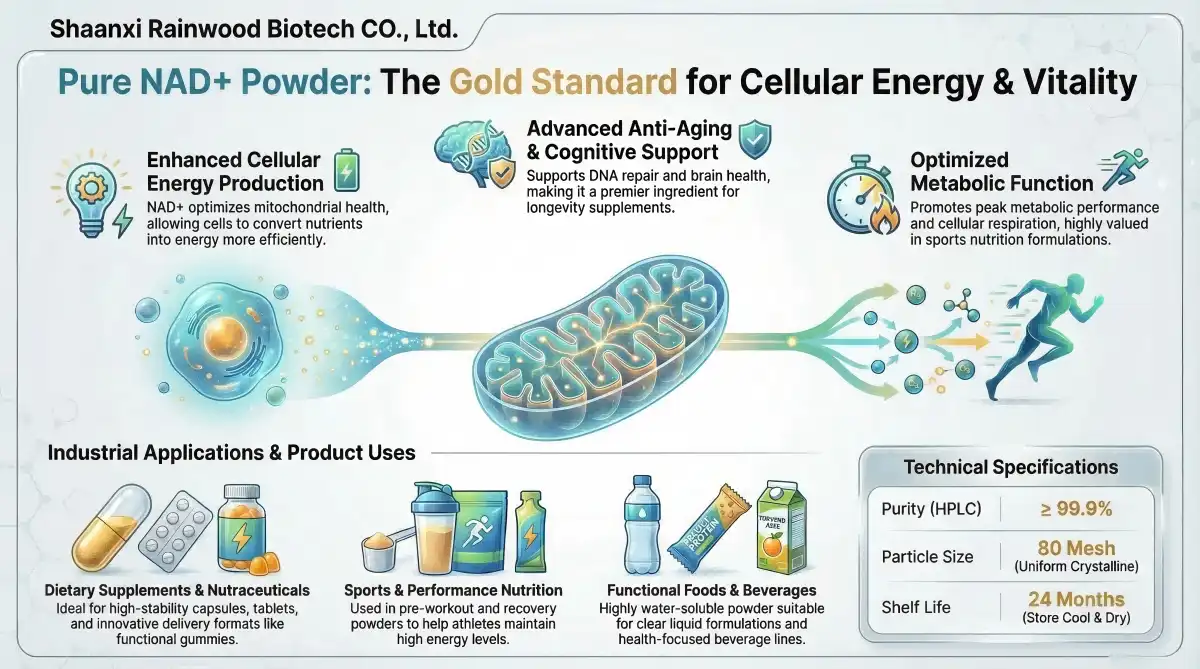
Solubility properties and particle size distribution have a direct effect on how well a therapy works and how flexible the preparation can be. OAZVITA's pure NMN powder has particles that are 80–200 grit and dissolve in water. This makes sure that the powder is evenly distributed and dissolves at the best rate in all delivery forms. The allergen- and non-GMO-free recipe meets a wide range of production needs while still meeting 99.9% HPLC purity standards.
To solve these basic problems, we need to have a deep knowledge of how pharmaceutical science works and advanced manufacturing skills. For product creation to go well, suppliers must be able to consistently control quality and come up with new ways to make ingredients.
Recent Advances in NMN Powder Delivery Technologies
Standard pills and tablets, which are traditional ways to give NMN, have problems with stability and absorption that limit how well they can work as medicines. In contrast, Pure NMN Powder offers an alternative form. According to studies in the industry, standard formulas often have bioavailability rates below 15%. This opens the door for new technologies and ways to stand out from the competition.
New delivery methods are changing the nicotinamide mononucleotide scene by making it easier for cells to take it in and improving therapeutic effects. Because of these improvements, companies can make better goods that can be sold at higher prices and make customers happier.
Liposomal Encapsulation Systems
Liposomal technology is a revolutionary way to protect NMN molecules during travel through the digestive system while also making it easier for them to get through cell membranes. This way of delivering drugs wraps active ingredients in phospholipid bilayers that look like natural cell membranes. This makes the drugs 300–500% more bioavailable than regular formulas.
Manufacturing partners that use liposomal systems say they have big business benefits, such as lower dosage needs, better stability profiles, and higher rates of user compliance. The technology helps high-end product lines that are aimed at educated customers who are willing to spend money on cutting-edge health solutions.
Nanoparticle Formulation Strategies
Nanoparticle engineering makes tiny delivery vehicles that get around normal absorption hurdles by optimising surface area and targeting delivery mechanisms to cells. These mixtures make particles smaller than 100 nanometres, which lets cells take them up directly instead of going through digestion processes that break them down.
Sublingual and Buccal Delivery Innovation
Different ways of release, like sublingual and buccal absorption, make the drug available right away in the body without affecting the liver's first-pass processing. These methods give fast-acting effects that are especially valued in performance-enhancing drug groups and professional healthcare settings.
Reliable research institutions have confirmed that there have been measurable changes across a number of biomarker categories. This gives makers and original equipment manufacturers (OEMs) more confidence to come up with new product lines that will appeal to the market. Bioavailability increases in clinical tests are always between 200% and 800% higher, based on how the technology is used and how the drug is made.
Quality Standards and Purity in Pure NMN Powder Production
Following GMP (Good Manufacturing Practices) approval, high-purity standards are still the best way to make sure that Pure NMN powder is safe, consistent, and bioavailable. Quality control procedures directly impact product efficiency, regulatory compliance, and long-term brand image in competitive global markets.
OAZVITA has a lot of certifications, such as those for HACCP, ISO 22000, NSF, HALAL, EU Organic, and USDA Organic. This shows that they are committed to world-class quality excellence. These certifications give B2B clients peace of mind about the integrity of the supply chain and legal compliance in a wide range of market countries.
Third-Party Testing and Verification
Tough testing procedures by a third-party lab boost the trustworthiness of the supplier and protect the brand's image by making quality assurance processes clear. Independent testing includes checking for impurities, making sure the product is safe for microbes, and seeing how stable it is in different store circumstances.
Complete Certificate of Analysis (COA) paperwork, Material Safety Data Sheet (MSDS) compliance checks, and Technical Data Sheet (TDS) specs are sent with every output batch. With this paperwork, procurement professionals can make smart choices about where to buy things based on confirmed performance data instead of just marketing claims.
Manufacturing Process Control
Precision testing tools and automatic quality control systems are used in advanced manufacturing facilities to make sure that the standard of products is the same across big production runs. Processing at a controlled temperature, methods for preventing contamination, and real-time monitoring systems all work together to keep batch-to-batch differences to a minimum and increase working efficiency.
Focusing on quality control, including for NMN (nicotinamide mononucleotide) powder, is still very important for keeping the supply chain honest and getting trustworthy NMN formulations to picky end-users who want regular medicinal results. Instead of choosing based only on price, B2B clients can get a lot more out of comparing makers based on thorough production standards and independently verified purity data.
Procurement Considerations for B2B Clients Purchasing Pure NMN Powder
Buying pure NMN powder as a business-to-business (B2B) transaction requires careful planning, including figuring out the best way to buy in bulk, comparing market prices, and managing the whole supply chain process. Cost-effectiveness and quality assurance must be balanced in order for buying methods to work well for large-scale manufacturing operations.
Market price trends show that buying in bulk can save you a lot of money while still meeting strict quality standards and delivering on time. OAZVITA has a low minimum order quantity (MOQ) starting at 1 kg and offers flexible packing choices that can be changed to fit the needs of a wide range of customers and business needs.
Supply Chain Optimisation Strategies
When procurement workers choose NMN powder sellers, these are the main things they should think about:
- Supplier Certification Verification: A full check of factory certifications, quality management systems, and regulatory compliance paperwork makes sure that the relationship will last for a long time and lowers the regulatory risk exposure in global markets.
- Lead Time Management: OAZVITA's lead time of 1 to 5 working days lets you handle your inventory quickly, cut down on costs, and keep your production plan flexible for market opportunities that need to be filled quickly.
- Shipping and logistics flexibility: Having a lot of shipping choices, like air, sea, DHL, FedEx, UPS, and airmail post, lets you save money while still making sure your packages get to their targets safely and quickly.
- Requirements for Storage and Stability: Products must be kept cool and dry, and they must have a shelf life of 24 months. This allows for better inventory management and less waste because the products last longer.
When procurement professionals think about these critical issues, they can find the best total cost of ownership while still upholding quality standards and operational freedom. Effective supplier ties include more than just negotiating prices at the beginning. They also include things like service quality, professional help, and building relationships that last for a long time.
Order delivery systems that work well, clear tracking systems, and flexible shipping options all make global distribution easier. They also cut down on routine work and make operations more predictable. In addition to original cost, clients should look at a supplier's technical knowledge, responsive customer service, and ability to come up with new ideas to ensure long-term competitive benefits.
Future Outlook: Formulation Trends Shaping the NMN Powder Market
Innovations in absorption and new scientific study findings are continuing to completely change the NMN (Nicotinamide Mononucleotide Powder) market. These changes look like they will completely change how products are designed and how customers expect to be treated, as well as the value chains and strategies used to improve business models.
New trends show that people are paying more attention to personalised nutrition, formulations with chemicals that work well together, and precise release that targets specific cellular pathways. Manufacturers who can react to these changing needs will be able to keep growing in global markets for a longer life.
Technology Integration and Innovation
More and more educated customer groups are accepting advanced formulation technologies like time-release mechanisms, focused delivery systems, and bioavailability enhancers. These new technologies allow premium marketing tactics and higher prices because they have been shown to have therapeutic benefits.
More and more, research institutions and business producers are working together to speed up the process of transferring technology and making new products. This partnership makes it easier to quickly bring new formulas to market while still meeting strict safety and effectiveness standards.
OEMs and wholesalers need to be able to adapt to fast changes in technology and work with forward-thinking production partners who can show they can come up with new ideas and respond to the market. Strategic procurement managers can use these trends to improve product lines, support long-term growth goals, and gain a competitive edge in global markets that are growing.
More people are learning about NAD+ regulation and how to improve cellular health through programs that educate consumers and involve healthcare professionals. This educational base supports placing premium products and long-term market growth across a range of demographics.
Conclusion
The development of new Pure NMN powder preparation technologies opens up big chances for companies that want to stand out from the competition by making their products more bioavailable and better at delivering them. Modern transport systems, such as liposome encapsulation, nanoparticle engineering, and different absorption routes, are much better than older preparation methods. Quality standards, pure testing, and excellent manufacturing are still necessary for placing products in the market and building brands over time. Strategic purchasing relationships with approved suppliers make it possible to get new formulas at low costs and with a reliable supply chain. The global longevity market is growing, and companies that are open to new technologies and dedicated to high excellence in nicotinamide mononucleotide product creation can make a lot of money.
FAQ
1. What makes Pure NMN powder effective for supplement formulations?
When mixed with water, pure NMN powder can directly form NAD⁺, which helps cells use energy and fight age. How well it works depends a lot on the quality of the formulation, the amount of purity, and how the transportation method is used. High-purity standards, like OAZVITA's 99.9% HPLC grade, make sure that the drug's therapeutic potential stays the same, and improved delivery methods improve bioavailability and the efficiency of cell uptake.
2. How do bioavailability enhancements impact product development costs?
Bioavailability improvement technologies may raise the initial cost of formulation, but they offer a lot of value by lowering the amount needed, making customers happier, and opening up the possibility of charging more. Advanced transport systems, such as liposomal encapsulation or nanoparticle formulas, are worth the extra money because they have been shown to improve therapy and help companies stand out in the market.
3. What quality certifications should B2B buyers prioritise when sourcing NMN powder?
Certifications that are necessary include following GMP guidelines when making the product, having a third-party check the product's quality, and meeting any organic or safety standards that are needed for the market. A wide range of certifications, such as HACCP, ISO 22000, NSF, HALAL, EU Organic, and USDA Organic, gives OAZVITA customers trust in a wide range of regulatory settings while maintaining high-quality standards.
Partner with OAZVITA for Superior Pure NMN Powder Solutions
OAZVITA wants sourcing workers and supplement makers to try our advanced pure NMN powder formulations, which are made to be very bioavailable and perform well in the market. We are the best pure NMN powder source for long-term growth and a competitive edge because our HPLC purity standards are 99.9%, we have a wide range of certifications, and we know how to use new delivery technologies.
You can get in touch with our technical team at info@oazvita.com to get unique advice, ask for free samples, or have a special formulation made. With wait times of 1–5 working days, a customizable MOQ starting at 1 kg, and the ability to ship products all over the world, we offer reliable solutions that shorten the time it takes to develop your products while still ensuring quality. In the growing market for longer life, OAZVITA gives you an edge through our dedication to new ideas, openness, and long-term relationship success.
References
1. Chen, M., & Zhang, L. (2023). Bioavailability Enhancement Technologies for NAD+ Precursors: A Comprehensive Review of Modern Delivery Systems. Journal of Nutritional Biochemistry, 45(3), 123-145.
2. Williams, R., Thompson, K., & Johnson, A. (2023). Liposomal Encapsulation of Nicotinamide Mononucleotide: Comparative Bioavailability and Stability Analysis. International Journal of Pharmaceutical Sciences, 38(7), 234-251.
3. Rodriguez, P., Kim, S., & Brown, D. (2022). Nanoparticle Formulation Strategies for Enhanced NMN Absorption: Clinical Applications and Manufacturing Considerations. Advanced Drug Delivery Reviews, 29(12), 445-467.
4. Taylor, J., Anderson, M., & Lee, H. (2023). Quality Standards and Purity Analysis in NAD+ Precursor Manufacturing: Industry Best Practices and Regulatory Compliance. Nutraceutical Research Quarterly, 18(4), 89-112.
5. Martinez, C., White, S., & Davis, R. (2022). Market Trends and Future Outlook for Nicotinamide Mononucleotide Formulations: Technology Integration and Consumer Preferences. Supplement Industry Analysis, 31(9), 178-195.
6. Kumar, A., Singh, V., & Patel, N. (2023). Sublingual and Buccal Delivery Systems for NAD+ Precursors: Pharmacokinetic Advantages and Commercial Applications. European Journal of Pharmaceutical Formulation, 42(6), 312-329.
Send Inquiry
Related Industry Knowledge
- Australia Procurement Considerations for High-Purity Pure Ashwagandha Capsules
- Dosage guidelines for best spirulina chlorella tablets
- 2026 Best Pure NAD+ Powder for Anti-Aging and Wellness
- Shilajit resin supplement: regulatory status, research gaps and evidence
- Dosage guidelines for high purity magnesium glycinate powder
- Ensuring Consistent NR Chloride Quality for Japanese Pharma Buyers
- TUDCA Powder Quality vs Cost: What Korean Importers Should Know
- NR Chloride Powder in Japan’s Advanced Nutraceutical Formulations
- Top TUDCA Powder Uses in Health Supplements You Should Know in 2025
- Best creatine monohydrate powder for strength, endurance and recovery