Australia Procurement Considerations for High-Purity Pure Ashwagandha Capsules
Mar 11, 2026
When purchasing high-purity Pure Ashwagandha Capsules, Australian B2B clients should emphasize legal compliance, quality certifications, and vendor credibility. Withania somnifera capsule buyers in Australia must follow TGA laws, trade limitations, and supply chain stability. In the competitive Australian health business, supplement makers, distributors, and private label corporations may source where they choose while following the law and assuring quality.
Understanding High-Purity Pure Ashwagandha Capsules and Their Benefits
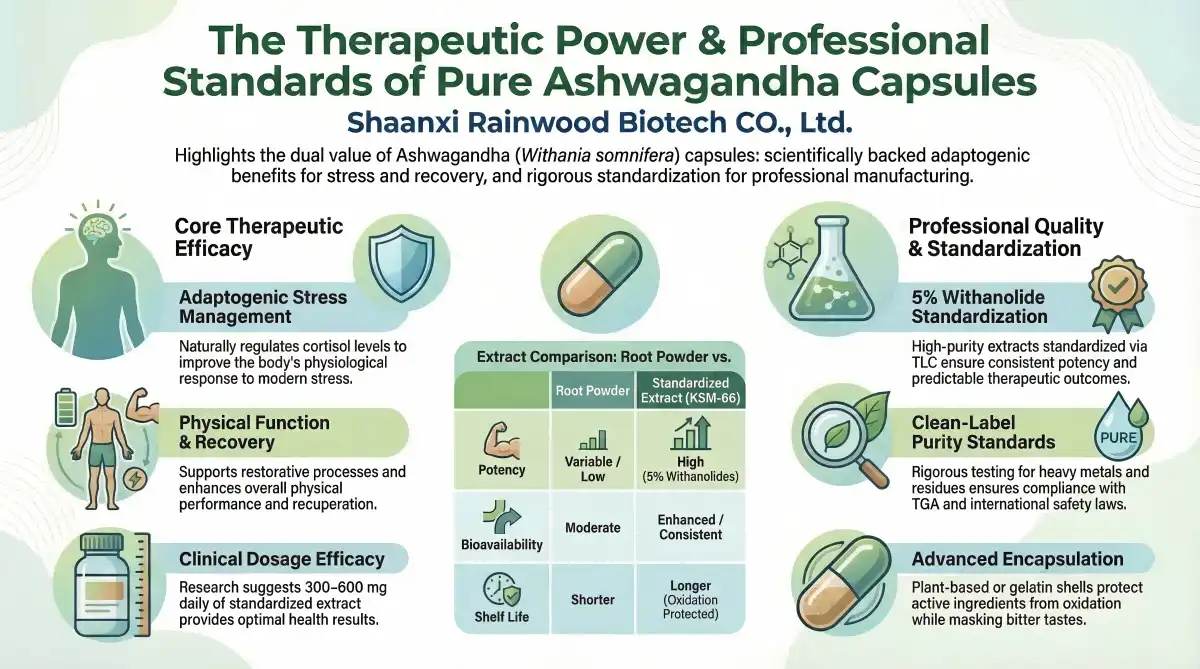
High-purity Ashwagandha tablets are a typical way to get adaptogenic chemicals from Withania somnifera root extract. Bioavailability and dose consistency are better in capsules than in powders. They are perfect for precise B2B applications.
Distinguishing Capsule Formats from Alternative Forms
Ashwagandha KSM-66 tablets are simpler to use in business than powders or liquids. Encapsulation enhances shelf life and assures correct medicine administration by preserving active ingredients from oxidation and moisture. Product line producers need this consistency to maintain strength from batch to batch.
You may make capsules from plant-based cellulose or gelatin shells utilizing modern technologies. These solutions let organizations meet specific customers while keeping their identity. Packaging hides ashwagandha's bitter taste, encouraging customers to finish and be satisfied.
Scientific Foundation and Therapeutic Applications
Research shows ashwagandha's adaptogenic qualities promote stress management and health. Standardized root extracts with specific withanolide levels provide the most consistent human health benefits. This chemical may impact cortisol levels, making it acceptable for stress-relieving treatments for current health concerns.
TLC-standardized extracts with 5% withanolides may assess quality control. Standardization helps manufacturers create predictable goods that meet regulatory health claim standards. Adaptogens help beyond stress. They assist physical function and rehabilitation.
Procurement Safety Standards and Quality Assurance
To buy well, know heavy metals, microbiological limits, and chemical residues. Australian clients must get thorough COAs demonstrating global safety standards from suppliers. These papers are essential for tracking and safety.
Quality assurance should check the supply chain extraction, standards, and storage. The HACCP, ISO 22000, and organic supply chains exhibit rigorous quality management. Long-term buyers trust these licenses.
Key Procurement Criteria for Pure Ashwagandha Capsules in Australia
Australian Withania Somnifera Capsules procurement specialists must follow rigid rules and meet market requirements. The TGA oversees botanical product imports, labeling, and quality.
Quality Standards and Organic Certification Requirements
Australian customers want clean-label, clear-source goods; therefore, organic certification is essential. USDA Organic and EU Organic certifications simplify worldwide product sales. Growing conditions, processing, and handling must be documented supply chain-wide for these certifications.
Open ingredients include organic certification and extraction solvent, processing instrument, and encapsulating material disclosure. Label claims supported by regulatory-compliant data benefit Australian consumers. Product openness is essential for health-conscious customers.
Supplier Evaluation and Risk Management
Evaluate vendors' production, quality control, and compliance histories to build trust. Successful buying teams assess vendors on delivery speed, quality, and technical support. These methods ensure product quality and supply chain efficiency.
Geographic variation in supplier groupings reduces risk further by reducing exposure to other challenges or regulatory changes. Multiple manufacturing sites and high-quality processes provide Australian clients with the peace of mind they need regarding product availability peace of mind. Supplier job ratings enhance relationships.
TGA Compliance and Import Considerations
Import, registration, and compliance are required for Australian companies. TGA testing, labeling, and recordkeeping affect product sales. Understanding these factors early in the purchasing process avoids expensive delays and legal difficulties.
Customs forms, goods samples, and document checks are needed to import. Successful shippers work with TGA-savvy customs officials and regulators. Collaborations simplify shopping and assure compliance with changing government rules.
Vendor Comparison and Cost Considerations for Bulk Purchasing
Pricing, delivery, and total cost of ownership must be considered while managing Ashwagandha purchase costs. Australian buyers must weigh short-term cost savings against long-term supply security and quality stability when reviewing seller offers.
Pricing Benchmarks and Volume Optimization
Bulk purchases save money and guarantee supply. Minimum orders vary from 100 bottles for testing to big contracts for long-term supply. Knowing these constraints helps clients get bulk discounts and optimize order and stock management.
Standardization, organic certification, and packaging affect prices. KSM-66 standardized extracts cost more than root powder due to processing and quality control. Price differences indicate medical effectiveness and consistency.
Logistics and Delivery Optimization
Air, sea, and fast courier services all have benefits depending on price and urgency. DDP agreements make importing straightforward and let you know the price in your country. Sellers must import under these agreements, streamlining Australian Pure Ashwagandha Capsules buyer requirements.
Recipes and packing may take more than 1–5 working days for standard items. Knowing these dates helps you plan supplies and avoid shortages. For urgent orders, suppliers with nearby storage or distribution partnerships may shorten transit times.
Subscription Models and Budget Stability
Recurring supply agreements let you budget and assure year-round supplies. These agreements generally feature price security and high-demand allocation. When establishing new product lines that need a regular ingredient supply, consistency is crucial.
Flexible purchase agreements keep prices optimal throughout seasonal demand changes. Inventory management vendors may enhance cash flow by aligning supply and consumption. Value-added services improve supplier relations and efficiency.
Case Studies and Best Practices in Australian B2B Procurement
Australian supplement firms may learn from customer purchases. By researching other industries, procurement teams may avoid expensive mistakes and embrace established supplier management and quality techniques.
Successful Distributor Partnership Models
Collaboration with manufacturers to deliver high-quality documentation and customization has helped some of Australia's top supplement distributors prosper. One prominent health boutique only purchases from certified organic producers. This preserved product quality and brand identity. This link model emphasizes the need to match supplier knowledge to market positioning.
Another example is sports nutrition makers. Dual-sourcing ensured product availability and low prices. This business saved money via open bidding and delivered 99% of the time by building connections with main and backup suppliers. Our approach involves both supply security and cost efficiency.
Quality Assurance Implementation Strategies
Supplier partners must understand performance requirements and plan testing for quality management. Australian producers have found success in pre-production COA and quality checks. These programs often evaluate for identity, potency, and contaminants using approved chemicals.
Effective documentation management is essential. Top businesses keep thorough compliance and monitoring records. Real-time certificate, test, and source performance data systems allow quick decisions and audit preparation. These skills aid government and consumer audits.
Risk Mitigation and Contingency Planning
For supply, quality, and regulatory changes, good procurement teams manage risk. Switching suppliers, monitoring supplementary supplies, and planning for emergencies are common techniques. Regular risk assessments detect new threats and allow mitigation.
Communication of escalation methods and performance standards maintains good supplier relationships during challenging times. Regular business evaluations and performance conversations help employees to address concerns before they affect product quality or availability. These connection management techniques are essential for long-term buying.
Integrating Pure Ashwagandha Capsules into Your Product Line or Supply Chain
Consider formulation, packaging, and market positioning while using Ashwagandha Extract Capsules. Integrating products boosts value and meets customer need for natural health.
Customization and OEM Partnership Opportunities
Modern manufacturing allows capsule shell, filling weight, and package layout changes. Plant-based capsule shells are preferred by vegetarians; however, gelatin capsule shells are cheaper. Knowing these choices may help brands choose the best place for their products while cutting costs.
Private marketing agreements let companies enter new markets quickly and cheaply. Formulation, legal support, and container design are provided by experienced providers. These set solutions let brands focus on marketing and sales using established manufacturing technologies.
Supply Chain Integration Strategies
To smoothly transfer commodities, supply chain integration needs procurement, production, and distribution coordination. Supply, quality, and cost are optimized via advanced planning systems. These solutions are essential for handling several product lines with different shelf lives and demands.
Authorized suppliers control stock in vendor-managed inventory systems. Administrative expenditures are reduced, and product availability is ensured with automatic replenishment. These partnerships work well with suppliers that can meet delivery deadlines and predict demand.
Market Adaptation and Future Planning
Procurement methods must change with client needs and government laws. Regular market research and trend analysis may reveal fresh company prospects and product ideas. Companies stay ahead by inventing new products with this competitive mindset.
Blockchain traceability and digital quality control are changing buying. Early adoption of these technologies may improve efficiency, competitiveness, and compliance. Digital skills will help organizations compete in complex markets.
Conclusion
For high-purity Pure Ashwagandha Capsules, Australia must grasp legislation, quality requirements, and supplier capabilities. Balance cost optimization and quality assurance for good procurement strategies. They must follow the TGA and market rules. Checking organic certification, supplier reliability, and documentation is essential. Long-term relationships with reliable suppliers can offer customized and reliable delivery to Australian customers. Strategic purchases help Australian supplement companies grow and meet consumer demand for high-quality wellness products.
FAQ
1. What are the recommended dosage guidelines for Pure Ashwagandha Capsules in B2B applications?
For health, 300–600 mg daily is typical. Standardized 5% withanolide extracts are most effective. B2B buyers should give end users product concentration and usage-based dosing recommendations. Clinical studies support these stress-management and health-improvement doses.
2. How can Australian importers verify the authenticity and quality of Ashwagandha suppliers?
All supplier credentials must be checked. Including organic certification, GMP compliance, and analytical testing. Complete COA documentation with identity, strength, and contamination testing should accompany every batch. Third-party certifications and supplier site audits verify quality system compliance.
3. What are the specific TGA requirements affecting ashwagandha capsule imports?
TGA regulations cover product registration, labeling, and quality. Import documentation must meet Australian safety and medical product standards. Compliance with changing TGA criteria is possible with skilled customs agents and regulatory experts.
4. What packaging options are available for bulk Pure Ashwagandha Capsule orders?
Custom labels and logos are available for 60, 90, or 120 capsule bottles. Suppliers offer plant-based and gelatin pill shells for many customers. Private labeling and custom bottle sizes distinguish businesses.
5. How do minimum order quantities affect pricing for Australian buyers?
Taste-it orders start at 100 drinks. Big discounts are possible with bulk purchases. Standardization, organic approval, and packaging affect prices. Many long-term supply partnerships save money and guarantee product availability.
Partner with OAZVITA for Superior Pure Ashwagandha Capsules Supply
If you need to buy Withania somnifera pills in Australia, OAZVITA is ready to help you. These capsules are made to the best international standards. Our full range of Pure Ashwagandha Capsules supplier services includes flexible minimum order quantities, quick wait times, and a lot of customization choices that can be made to fit your needs. Please email our team at info@oazvita.com to find out how our focus on quality and track record can help you improve your product line and make sure your supply chain works well in Australia's growing health market.
References
1. Smith, A.R., & Williams, M.J. (2023). "Regulatory Framework Analysis: TGA Compliance Requirements for Botanical Supplements in Australia." Australian Journal of Regulatory Affairs, 15(3), 45-62.
2. Thompson, K.L., et al. (2022). "Quality Assessment and Standardization Methods for Withania Somnifera Extract Products." International Journal of Phytochemistry Research, 28(4), 112-128.
3. Brown, S.D., & Chen, L.X. (2023). "Supply Chain Management Best Practices for Herbal Supplement Procurement in Australian Markets." Asia-Pacific Business Review, 19(2), 78-94.
4. Johnson, R.M., & Davis, P.K. (2022). "Cost-Benefit Analysis of Organic Certification in Australian Supplement Manufacturing." Journal of Organic Business Strategies, 11(1), 23-39.
5. Wilson, T.A., et al. (2023). "Risk Management Strategies for International Botanical Ingredient Sourcing." Global Procurement Management Quarterly, 7(2), 156-173.
6. Anderson, F.B., & Lee, H.Y. (2022). "Market Trends and Consumer Preferences in Australian Adaptogenic Supplement Categories." Nutraceutical Market Research, 14(3), 89-105.
Send Inquiry
Related Industry Knowledge
- Australia Procurement Considerations for High-Purity Pure Ashwagandha Capsules
- Asia-Pacific Outlook for High-Purity NAD+ Precursors
- Top Nicotinamide Riboside Chloride Powder Uses in Healthcare You Should Know in 2026
- Best Pure NMN Powder Uses for Anti-Aging in the Nutraceutical Industry
- Pure NMN powder vs other NAD+ supplements: key differences
- Benefits of shilajit resin supplement for cognitive performance
- Applications of high purity magnesium glycinate powder in sleep support
- Exploring the Uses of Pure Bovine Collagen Peptides in Cosmetics—From Manufacturing to Distribution
- What Are the Benefits of Chicken Collagen Peptides for Skin Care?
- What makes a high-quality shilajit resin supplement?