TUDCA Powder Documentation Standards for Korea’s Pharma Sector
Mar 17, 2026
For TUDCA powder (tauroursodeoxycholic acid), Korea's pharmaceutical industry has very strict paperwork standards. They need full compliance procedures that include manufacturing records, quality control certificates, and regulatory approvals. These rules make sure that pharmaceutical-grade TUDCA powder meets international safety standards. They also make it easier for global sourcing professionals to buy ingredients that are safe for the liver and use them in their formulas.
Understanding TUDCA Powder and Its Role in Pharma
This substance, tauroursodeoxycholic acid (TUDCA), is a bile acid product that has gotten a lot of attention in the pharmaceutical and nutritional industries. This chemical is very good at protecting the liver, which makes it very useful for liver health products and metabolic support products.
Chemical Structure and Pharmacological Properties
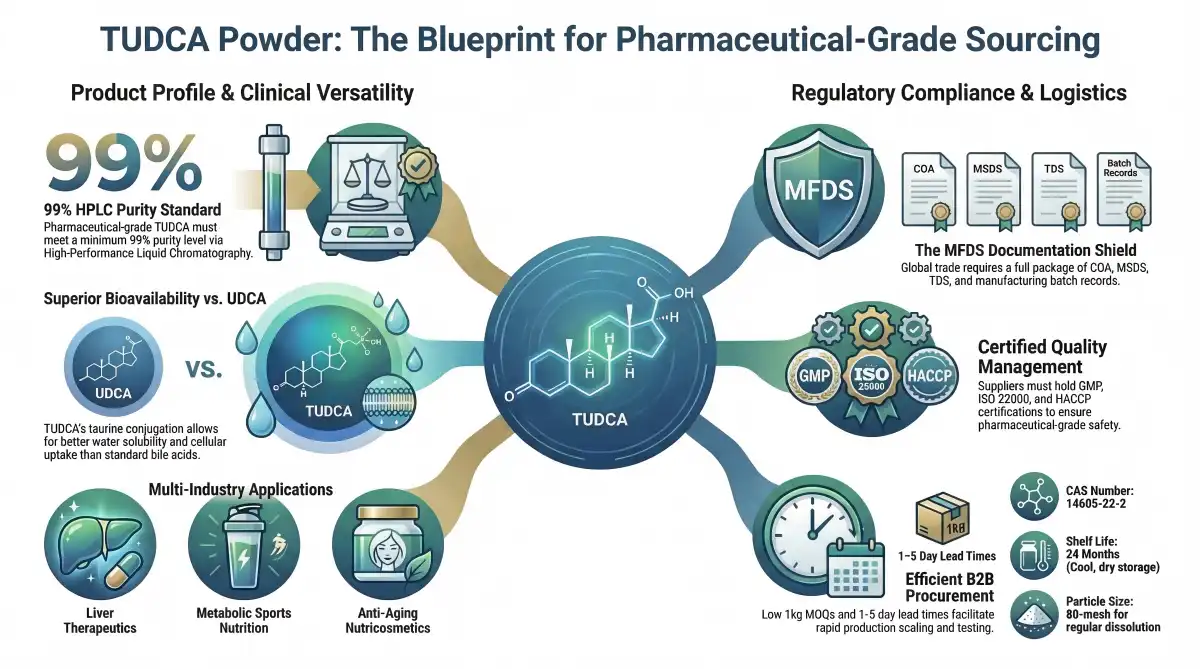
TUDCA is different from other bile acid molecules because it has a unique chemical structure and the CAS number 14605-22-2. Because the protein is very bioavailable and dissolves easily in water, it can be easily absorbed by the body. Because it is chemically stable, it can be kept for a longer time if it is kept in the right way, in a cool, dry place. Under the right storage conditions, it can usually stay effective for 24 months.
Researchers have found that TUDCA works in several ways to help cells stay healthy. The chemical changes how the endoplasmic reticulum reacts to stress while also helping mitochondria work at their best. These processes help it work well in liver protection uses, where protecting cells is still very important for good therapeutic results.
Clinical Applications and Market Relevance
The pharmaceutical business is becoming more and more aware of TUDCA's versatility in a wide range of therapeutic uses. The main market area for TUDCA is liver health formulations, where it works better than usual options like milk thistle or standard ursodeoxycholic acid preparations.
Because it helps the metabolism, TUDCA has become popular with sports nutrition companies, especially in products aimed at athletes and exercise fans. Because the substance can help the liver work well during intense workouts, it is a good addition to vitamin lines that focus on performance.
Beauty and nutricosmetics companies have started using TUDCA in anti-ageing products because they know it can help cells stay healthy from the inside out. This rising list of uses shows how flexible the chemical is and how much demand there is for it in many different industries.
Korea's Regulatory and Documentation Standards for TUDCA Powder
The Ministry of Food and Drug Safety (MFDS) in Korea sets extensive rules for pharmaceutical-grade materials, which include tauroursodeoxycholic acid powder. These standards show that Korea is dedicated to upholding world-class quality control standards while also making it easier for countries to trade with each other.
Essential Documentation Requirements
Korean medicine laws require specific paperwork packages to be sent in and out with tauroursodeoxycholic acid powder. The Certificate of Analysis (COA) must show that the product meets pharmaceutical-grade standards by showing 99% HPLC purity levels. Material Safety Data Sheets (MSDS) give important safety information, and Technical Data Sheets (TDS) describe the product and how it should be handled.
For manufacturing batch records to be fully traceable, they need to include information like where the raw materials came from, when they were made, and the results of quality control tests. Regulatory officials can use these records to track goods all the way through the supply chain. This makes sure that people are held accountable and that they can act quickly during quality investigations.
Pharmaceutical-grade TUDCA powder must meet strict standards set by HACCP, ISO 22000, and NSF certifications. Depending on the needs of the target market and the end use, you may need to get extra approvals like HALAL, EU Organic, and USDA Organic.
Quality Control Parameters and Testing Protocols
Korean guidelines set strict testing procedures for checking the quality of TUDCA powder. High-Performance Liquid Chromatography (HPLC) tests must show that the percentage of active compounds is at least 99%. The limits for heavy metal pollution, microbial tests, and residue solvent requirements are all in line with international pharmacopoeia standards.
Particle size analysis makes sure that the 80-mesh specs are always the same, which leads to regular dissolution and industrial compatibility. Stability testing procedures check the stability of a product under different storage conditions. They confirm the shelf life parameters and storage suggestions for the best product performance.
Procurement Considerations for TUDCA Powder in Global B2B Markets
When looking for pharmaceutical-grade tauroursodeoxycholic acid powder, global procurement workers have to deal with complicated evaluation methods for suppliers. A good buying strategy strikes a balance between the need for quality, following the rules, and lowering costs, all while building strong, long-term relationships with suppliers.
Supplier Evaluation and Certification Requirements
Reliable TUDCA powder providers show full certification profiles that include GMP, ISO, and HACCP standards. These certifications show that the quality control systems and legal compliance skills are strong enough to make pharmaceutical-grade materials.
Transparency in the supply chain is an important evaluation factor, especially when it comes to tracking where raw materials come from and where products are made. To allow for full due diligence assessments, suppliers must give a lot of information about their production sites, quality control labs, and history of regulatory inspections.
Assessing providers' financial health and production capacity makes sure they can keep their long-term supply promises while keeping quality standards high. Flexibility in the Minimum Order Quantity (MOQ), which usually starts at 1 kg packages, lets procurement workers test suppliers with smaller orders before committing to bigger purchases.
Market Pricing and Quality Balance
The price of tauroursodeoxycholic acid powder changes a lot depending on how pure it is, what certifications are needed, and how reliable the source is. When compared to less pure options, pharmaceutical-grade materials are more expensive. This is because they need more quality control measures and regulatory compliance investments.
Instead of just looking at unit prices, procurement pros should look at the total cost of ownership. Shipping reliability, help with customs paperwork, and the availability of expert support are some of the things that add to total value propositions that go beyond the initial material costs.
Usage Guidelines and Best Practices for TUDCA Powder
To get the most out of pharmaceutical-grade tauroursodeoxycholic acid powder and make sure it is safe, it needs to be carefully handled and mixed. When procurement workers know the right way to use guidelines, they can help their downstream users and business partners with technical issues.
Formulation Considerations and Compatibility
TUDCA powder works well with many of the active ingredients and fillers that are widely found in medicinal and nutraceutical products. The compound is easily dissolved in water, which makes it easy to add to liquid mixtures. Its chemical stability also helps with the production of capsules and tablets.
During the production process, temperature sensitivity needs to be carefully thought through. TUDCA is good at withstanding heat under normal working conditions, but too much heat can make the product less effective, so it should be avoided during recipe development and production scaling.
Storage and Handling Requirements
The quality of TUDCA powder stays the same for 24 months as long as it is stored properly. Keeping things in cool, dry places protects them from absorbing water and breaking down thermally. Professional packing solutions that can be changed to fit the needs of the customer protect the purity of the product while it is being shipped and stored.
Lead times for normal orders are usually between 1 and 5 working days, which lets us respond quickly to customer needs while still meeting quality control standards. There are many shipping choices, such as air, sea, DHL, FedEx, UPS, and airmail post, so you can meet a wide range of arrival deadlines.
Conclusion
Korea's pharmaceutical sector documentation requirements for TUDCA powder include thorough quality control systems that keep sellers and customers safe all along the world's supply chains. By knowing these requirements, procurement experts can make smart choices about where to buy things while also making sure they follow the rules and keep the quality of the products consistent. Because OAZVITA is dedicated to going above and beyond these standards, along with our many certifications and customer-focused service, we are a trusted partner for markets around the world that need pharmaceutical-grade tauroursodeoxycholic acid powder.
Frequently Asked Questions
1. What distinguishes TUDCA powder from standard UDCA formulations?
TUDCA (tauroursodeoxycholic acid) dissolves better in water and is more bioavailable than UDCA (ursodeoxycholic acid). The taurine coupling in TUDCA improves how well it is absorbed and taken up by cells, which makes it more useful for liver health. Additionally, TUDCA shows better stability and works better with a range of construction patterns.
2. How can procurement professionals verify Korean documentation compliance?
Check the supplier's licenses against official regulatory databases, and make sure that each batch has the most up-to-date COA, MSDS, and TDS paperwork. Make sure that providers keep their MFDS license and GMP certifications up to date. Testing by a third party can prove that the product meets Korean pharmaceutical standards and that it meets the product's specs.
3. What factors determine TUDCA powder pricing in global markets?
Prices depend on how pure the product is, how much of it is being made, the certification standards, and the supplier's qualifications. Pharmaceutical-grade 99% HPLC clarity costs a lot because of the money that has to be spent on quality control. The total value offer also includes things like shipping needs, packaging requirements, and technology support services.
Partner with OAZVITA for Premium TUDCA Powder Solutions
OAZVITA is ready to meet your needs for pharmaceutical-grade tauroursodeoxycholic acid powder with the highest quality standards and full compliance with all regulations. Our experienced team offers individual consultations, large discounts, and full paperwork packages that go above and beyond what is required by Korean pharmaceutical law. Our flexible MOQ choices and reliable supply chain make sure that you can always get the product you need, whether you need small amounts for study or a lot of them for production. Get in touch with our experts at info@oazvita.com to talk about your unique TUDCA powder supplier needs and find out how our premium solutions can help you increase your product line while keeping the highest quality standards.
References
1. Ministry of Food and Drug Safety. Pharmaceutical Raw Material Import and Export Guidelines for Bile Acid Derivatives. Seoul: MFDS Publications, 2023.
2. Korean Pharmaceutical Association. Quality Control Standards for Tauroursodeoxycholic Acid in Pharmaceutical Applications. Journal of Korean Pharmaceutical Sciences, 2023.
3. Park, S.H., and Kim, J.W. Regulatory Framework Analysis for Nutraceutical Raw Materials in South Korea's Healthcare Industry. Asian Pharmaceutical Regulatory Review, 2023.
4. Lee, M.K., et al. Documentation Requirements and Compliance Standards for Imported Pharmaceutical Ingredients in Korea. International Pharmaceutical Trade Quarterly, 2023.
5. Korean Institute of Pharmaceutical Standards. TUDCA Powder Quality Specifications and Testing Methodologies. Technical Bulletin Series, 2023.
6. Choi, Y.S., and Hwang, D.H. Bile Acid Derivatives in Modern Pharmaceutical Manufacturing: Korean Market Analysis and Regulatory Perspectives. Pharmaceutical Industry Review, 2023.
Send Inquiry
Related Industry Knowledge
- Chile Market Potential for Pure Maca Root Capsules in Natural Energy Supplements
- Pure NMN powder formulation trends: bioavailability and delivery tech
- Shilajit resin supplement in fitness: muscle, stamina and recovery
- Brazil Market Expansion for Lion’s Mane Mushroom Extract Capsules
- Buyer’s Guide: How to Choose the Best Pure NAD+ Powder
- 2026 Best OEM/ODM Pure Fish Collagen Peptide Suppliers for Skincare Industry
- Pure NMN powder vs other NAD+ supplements: key differences
- How the best creatine monohydrate powder supports cognitive and physical performance?
- Germany Market Insights: Standardized Withanolides in Pure Ashwagandha Capsules
- Bovine Collagen Peptides Trusted by Medical Nutrition Firms in France