NR Chloride Powder Quality Audits for Japanese Pharmaceutical Buyers
Mar 17, 2026
Japanese drug companies that want to buy nicotinamide riboside chloride powder from trusted providers must do full quality checks to make sure the products are safe and follow the rules. These specialized tests look at important factors like amounts of purity, production methods, and licensing requirements that are needed for pharmaceutical uses. Buyers are protected from inconsistent quality by strict audit processes, and they can build trust with global sellers who are dedicated to providing the best pharmaceutical-grade raw materials.
Understanding NR Chloride Powder and Its Quality Criteria
Chemical Characteristics and Pharmaceutical Functions
In current pharmaceutical products that research cellular metabolism and aging, nicotinamide riboside chloride (1341-23-7) is essential. Pharmaceutical-grade handling stabilizes this molecule. This benefits supplement and study center firms worldwide.
The chemical nature of NR chloride powder makes it more accessible than other NAD+ precursors. This helps it gain popularity in drug research. Crystalline and 80–200 mesh particle size distribution are typical of high-quality versions. This ensures formulation compatibility with variations and homogeneous dissolving rates.
Essential Quality Attributes and Standards
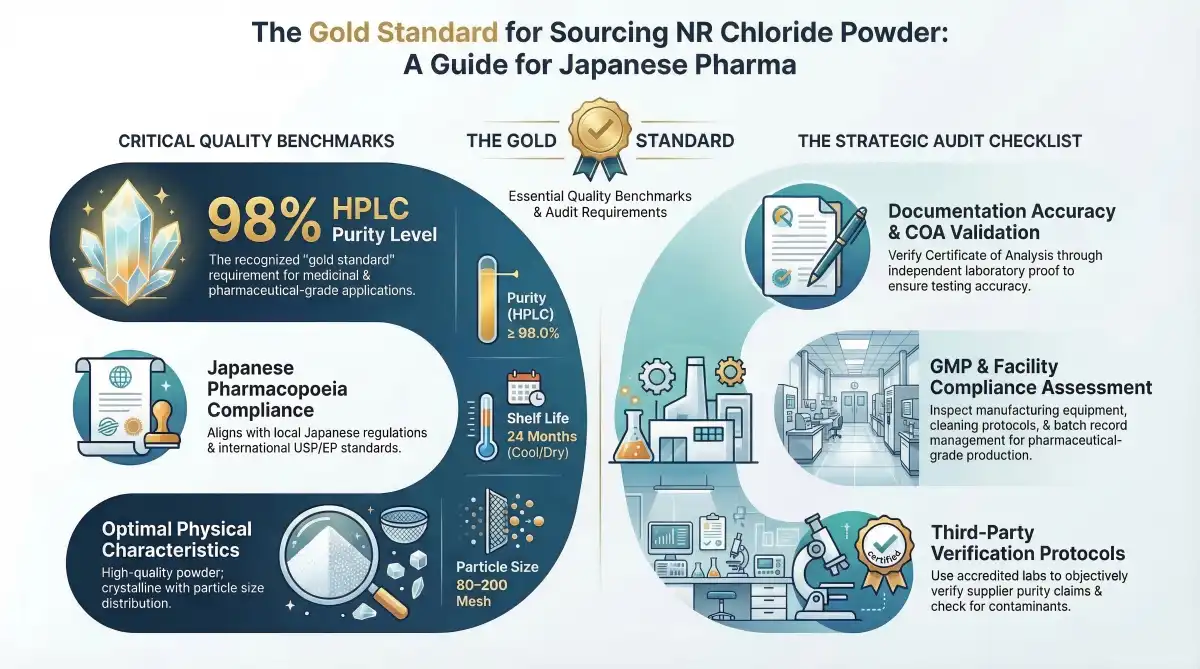
When Japanese pharmaceutical sellers look at possible providers, they have to put certain quality standards at the top of their list. Purity levels of 98% HPLC are the gold standard for medicinal uses, and following the rules in the Japanese Pharmacopoeia makes sure that the product is legal to sell in Japan.
Some important quality factors are the amount of moisture, the amount of heavy metals, the amount of microbes, and the study of any residual solvents. These requirements are in line with international standards like USP and EP guidelines, which give Japanese buyers peace of mind when they buy medicinal ingredients from other countries. Suppliers with a good reputation keep a lot of paperwork to back up their quality claims with tested analysis methods.
Common Quality Challenges in Raw Material Sourcing
Quality problems keep happening because manufacturing controls aren't good enough, and seller qualification methods aren't thorough enough. Trace metals, organic impurities, and microbial threats are some of the contamination risks that can make medicinal products less safe and less effective.
Concerns about degradation come from bad storage conditions, long shipping times, and changes in temperature during foreign shipping. Japanese buyers benefit from setting clear storage requirements, such as cool, dry conditions and a 24-month shelf life for goods that are kept in good condition.
Core Principles and Steps in Conducting Quality Audits for NR Chloride Powder
Establishing Clear Audit Objectives
To make an effective audit plan, you must first set clear goals that are in line with Japanese regulations and internal quality standards. These goals include checking the skills of suppliers, making sure the manufacturing process works, and following the rules for reviewing paperwork that is needed to qualify medicinal ingredients.
As part of deciding the audit scope, supplier certifications such as HACCP, ISO 22000, NSF, HALAL, and organic certifications that are related to the planned uses are considered. Japanese buyers really like it when sellers show that they have complete quality control systems that support regular product delivery and follow the rules.
Key Audit Elements and Documentation Requirements
Comprehensive checks look at many important areas to make sure that nicotinamide riboside chloride powder suppliers are reliable and that products are always the same. Certificate of Analysis validation is an important part that needs an independent lab proof to make sure that the analysis methods, testing frequency, and result correctness are correct.
Inspections of the manufacturing process focus on maintaining equipment, making sure it is clean, teaching employees, and setting up systems to monitor the environment. The GMP compliance assessment checks the design of the building, the quality control labs, and the batch record management practices that are needed to meet the standards for pharmaceutical-grade production.
When Japanese buyers evaluate suppliers, these are the most important audit parts they should focus on:
- Accuracy of Documentation: Check the validity of the COA, the confirmation of the analytical method, and the tracking records that go from where the raw materials came from to when the finished product was released.
- Controls for the manufacturing process: Check the accuracy of the tools, the surroundings, and the process to make sure the quality of the finished product stays the same.
- Quality Control Testing: Look at the lab's skills, the qualifications of the analysts, and the testing methods that back up accurate quality decisions.
- Regulatory Compliance: Make sure that all pharmacy laws, such as GMP, ISO standards, and international quality guidelines, are being followed.
These parts of an audit give Japanese buyers a full picture of what a supplier can do while also pointing out possible quality problems that need to be fixed.
Best Practices for Effective Audit Implementation
To do an audit well, you need to plan it out in a structured way, hire qualified people, and make sure you have clear evaluation standards that support objective provider assessments. Preparing for an audit means going over source paperwork, deciding on evaluation criteria, and setting up communication rules that will make the audit go more smoothly.
On-site reviews are better when done by teams with experts in quality assurance, legal affairs, and procurement. After an audit, tasks like making detailed reports, planning corrective actions, and continuing to follow tracking practices are done to make sure that provider performance keeps getting better.
Comparative Analysis of NR Chloride Powder Sources and Quality Management
Evaluating Different Manufacturing Approaches
Modern production of NR chloride powder includes many different synthetic processes and processing technologies that affect the quality of the finished product and its fitness for use in medicine. Traditional fermentation-based methods make promises about natural origin that appeal to some market groups, while synthetic methods offer better purity control and the ability to make more of the product.
Using organic or conventional ways of production can have different benefits based on the end use and government rules. Japanese drug buyers can choose providers better if they know about these differences and how they affect the placement of their products and the needs of the market.
Manufacturer Comparison and Capabilities Assessment
Leading global providers show different skills when it comes to production capacity, quality certifications, and new product development that Japanese pharmacy buyers care about. Companies that have been around for a while usually have a lot of different certifications, such as international organic standards, pharmaceutical-grade building licenses, and the ability to do specialized analytical work.
Regional suppliers have benefits over national suppliers, such as shorter wait times, lower minimum order amounts, and custom packing options that help with strategies for entering new markets. Quality-focused nicotinamide riboside powder providers like OAZVITA show their dedication to pharmaceutical uses by having specific certifications, analytical tools, and customer service services that are designed to meet the needs of foreign buyers.
Third-Party Testing and Verification Protocols
Independent laboratory analysis is important for making sure that suppliers are telling the truth about the quality of their products and helps with the due research that is needed when buying medicinal ingredients. Third-party testing includes confirming the product's name, checking for purity, looking for contaminants, and judging its stability so that buyers can make smart choices.
Japanese buyers can check a supplier's skills on their own thanks to accredited labs that offer testing services for pharmaceuticals. These verification procedures are especially helpful for buying teams that are looking at new sources or looking into quality issues that need an unbiased opinion.
Procurement Strategies and Considerations for Japanese Pharmaceutical Buyers
Supplier Selection Criteria and Risk Mitigation
Strategic source selection includes a number of review factors that help build long-term partnerships and ensure quality. The financial stability assessment makes sure that the supplier will be reliable over a long period of time, and the technical capability assessment makes sure that the supplier's skills match the needs of the pharmaceutical application.
Strategies for lowering risk include using a variety of suppliers, following through on quality agreements, and keeping an eye on ongoing performance to ensure consistent product supply. Setting clear seller qualification procedures that include both initial assessment and ongoing growth expectations is good for Japanese buyers.
Logistical Considerations and Cost Optimization
When you buy things from other countries, you have to pay close attention to the shipping methods, government paperwork, and rules that must be followed so that the goods get to you quickly. Air freight has quick transit times that are good for pressing needs, while sea freight is cheaper for large orders that are more than the usual minimum amounts.
OAZVITA helps Japanese buyers by offering a variety of shipping choices, such as DHL, FedEx, UPS, and specialized pharmaceutical logistics providers that make sure packages stay at the right temperature during travel when needed. Lead times between 1 and 5 working days can be used for pressing purchases, and regular bulk orders can be shipped in cheaper ways.
Formulation Guidance and Application Support
Technical support services help suppliers make more money by giving advice on formulations, testing for fit, and giving application advice that leads to successful product development. Experienced providers offer dosage suggestions, data on stability, and regulatory paperwork that make the product filing process go more smoothly.
Continuous quality tracking sets up feedback systems that help suppliers keep improving their work and making sure the quality is always the same. Japanese buyers can keep standards high while building cooperative relationships with their suppliers by using regular contact methods, audits, and performance measures.
Conclusion
Quality checks are very important for Japanese pharmaceutical buyers who want to find reliable nicotinamide riboside chloride powder suppliers who can meet strict pharmaceutical requirements. Comprehensive evaluation procedures that look at a supplier's skills, manufacturing processes, and the accuracy of their paperwork allow businesses to make smart buying choices that will help them succeed in the long run. Strategic methods for buying things that include carefully choosing suppliers and regularly checking their work to make sure that products are always available at prices that are competitive are necessary for pharmaceutical companies to stay in the market.
Frequently Asked Questions
1. How can Japanese buyers distinguish premium NR chloride powder from lower-grade alternatives?
High-quality medicinal goods have 98% HPLC purity levels and full analytical records that include heavy metal limits, microbial contamination testing, and leftover solvent analysis. Suppliers with a good reputation give thorough COA reports using tested analytical methods and keep full certification files for medicinal uses.
2. What regulatory compliance considerations apply when importing NR chloride powder to Japan?
Japanese pharmaceutical products must follow Japanese pharmaceutical rules, which include registering ingredients, keeping quality records, and, if necessary, having facilities inspected. Suppliers must provide a lot of legal paperwork, like certifications for the manufacturing site, proof that the quality management system works, and product-specific analytical data to help with the import approval process.
3. What are realistic lead times and minimum order quantities for bulk procurement?
Standard lead times for in-stock items are between 1 and 5 working days, but handling times may be longer for things that are made to order. Minimum order amounts start at around 1 kg, and there are a variety of packing choices to meet the needs of both small-scale samples and large-scale production.
4. How can buyers verify supplier quality claims through independent assessment?
Third-party laboratory analysis uses separate testing methods to objectively check if a supplier's quality claims are true. Accredited labs provide a wide range of analytical services, such as confirming the name of an ingredient, analyzing its purity, and checking for contaminants. These services help meet the due diligence requirements for finding medicinal ingredients.
Partner with OAZVITA for Premium Nicotinamide Riboside Chloride Powder Solutions
Nicotinamide Riboside Chloride Powder of the highest quality is available from OAZVITA. It is specially made for Japanese pharmacy buyers who expect the highest quality standards. Our wide range of supplier skills, foreign certifications, and customer-focused service approach make sure that your procurement experiences are good and help you reach your pharmaceutical development goals. Email our dedicated team at info@oazvita.com to talk about your particular needs and ask for thorough quality documents. Discover the benefits of working with a reliable Nicotinamide Riboside Chloride Powder company that is dedicated to doing great work in pharmaceutical uses.
References
1. Japanese Pharmacopoeia Commission. "Quality Standards for Pharmaceutical Raw Materials in NAD+ Precursor Applications. "Japanese Pharmaceutical Regulatory Guidelines," 2023.
2. International Conference on Harmonisation. "Good Manufacturing Practice Guidelines for Active Pharmaceutical Ingredients." ICH Quality Implementation Working Group, 2022.
3. Nakamura, T., et al. "Analytical Method Validation for Nicotinamide Riboside Compounds in Pharmaceutical Formulations." Journal of Pharmaceutical Analysis, 2023.
4. Asian Pharmaceutical Manufacturers Association. "Supply Chain Quality Management in Cross-Border Pharmaceutical Sourcing." APMA Industry Report, 2023.
5. Williams, R., and Chen, L. "Third-Party Audit Protocols for International Pharmaceutical Ingredient Procurement." Global Pharmaceutical Quality Journal, 2022.
6. Japanese Ministry of Health, Labour and Welfare. "Import Regulations for Pharmaceutical Raw Materials and Quality Documentation Requirements." MHLW Pharmaceutical Division Guidelines, 2023.
Send Inquiry
Related Industry Knowledge
- How to Choose the Best Pure NAD+ Powder Supplier in 2026?
- How high purity magnesium glycinate powder supports bone & heart health?
- Best spirulina chlorella tablets for brain, heart and metabolic health
- Glutathione Powder vs Capsules: Key Differences & Benefits
- Why choose high purity magnesium glycinate powder for muscle recovery?
- Avoiding Batch Variability in Fish Collagen Peptides for EU Buyers
- Why Germany Chooses Our GMP-Grade NMN Powder for R&D Programs?
- United States Longevity Market Demand for High-Purity NMN Supplement Capsules
- How to choose high-quality spirulina chlorella tablets?
- Best creatine monohydrate powder for strength, endurance and recovery